Patologia Molecular
Transcript of Patologia Molecular

Harvy Mauricio Velasco PGenética Humana
UNAL

1* 1015 células adultas17 2*1017 divisiones
celularesó6 *109 nucleótidos
para cada división l lcelular
1*10 -6 -8 Eficiencia de l DNA li la DNA polimerasa

Proliferación, Noxas externasDivisión yapoptosis
Noxas externas
Oncogenes y Genes
Reparación delDNA
supresores


UN GEN SE UN GEN SE ENCUENTRA ENCUENTRA AFECTADO

GENERADAS POR LA EVOLUCION
ERRORES EN LA REPLICACION
DNA POLIMERASANOVEDADADAPTACION
DNA POLIMERASA
TAMAÑO DE DNA3 000 MILLONES DE pb
ENFERMEDADMUERTE
3.000 MILLONES DE pb
AGRESION DIRECTA A DNADEGRADACION INTERNA DEGRADACION INTERNA DE NUCLEOTIDOS

MUTACIONMUTACIONCualquier cambio en el material genético(DNA
POLIMORFISMOV i ió étimaterial genético(DNA
nuclear y mitocondrial), que se herede a las
Variación genética que tiene más de
i tgeneraciones siguientes. una variante alélica en una
i DNAsecuencia DNAFrecuencia >1%

MUTACIONES GERMINALES
MUTACIONES SOMATICASM t ió GERMINALES
El cambio en el DNA se da lí t l
Mutación que no compromete líneas germinalesen línea parental en
celulas germinales y esta mutación se HEREDA
germinalesEn un tejido especifico
mutación se HEREDA

GERMINALES SOMATICAS
INDUCIDAS POR ELAMBIENTE O
EN EL LABORATORIOESPONTANEAS

A GRAN ESCALA O MUTACIONES
A PEQUEÑA ESCALA O CITOGENETICAMENTE MUTACIONES
CITOGENETICAMENTE VISIBLES
CITOGENETICAMENTE INVISIBLES
VISIBLES

EXISTE ALTERACION DE ALGUNOS DE LOS COMPONENTES DEL MATERIAL GENETICO O EN EL FLUJO DE INFORMACION DEL DNA HACIA EL FLUJO DE INFORMACION DEL DNA HACIA PROTEINAS
ERROR EN LA EXPRESION DE LA INFORMACION GENETICAGENETICA

TIPO DEFINICIONSUSTITUCION Reemplazo de una(s) base(s)SUSTITUCION Reemplazo de una(s) base(s)
por otra(s)
DELECION P did li i ió dDELECION Perdida o eliminación de uno o más nucleótidos
INSERCION Uno o más nucleótidos están adicionados en una secuencia
–Transposición con duplicaciónde material genético–Transposición sin duplicación(translocación)

MUTACIONES
Sustitución de Inserciones Delecionesbases
Transiciones Transversiones
1 o pocos nucleótidos
1 o pocos nucleótidos
Sustitución i ó i ( t )
Expansión de i l
Grandes delecionessinónima (neutra)
y no sinónimatripletas deleciones
Conversión génica, múltiple sustitución de
Otras grandes inserciones: elementos
ibl
En el marco de lectura
bases transponibles


Transiciones: Pirimidina por
Transversiones:Purina por pirimidina o p
pirimidina (C-T)Más comunes (ISLAS
p pviceversa
(CpG)
Purina por purina (A-G)G)

A C
G TG T


SINÓNIMAS (SILENCIOSAS): Ó(SILENCIOSAS):NeutralNo cambia secuencia
NO SINÓNIMASAltera secuenciaNo cambia secuencia
a.a.En DNA no codificante
á ú
TIPOSDeletéreasSin efectoMás común en DNA
codificante En DNA codificante
Sin efecto Efecto benéfico
En DNA codificante cambia el A.A. en posición 3 de codón

Mayor frecuencia Transversiones que transicionesMayor frecuencia en secuencias no codificantesCualquier secuencia puede ptener sustituciones No
sinónimas
Sinonimias
Sinónimas

Algunas sustituciones en secuencias no codificantes afectan la expresión

SUST
MUTAC
ON
enerac
ón
de
ás
ca
de
a
MUTAC
ON
erac
ón
de
o
CONS
p
azado
qu
m
NO
CON
qu
m
camen
eSUSTITUCIONES
TUC
ONES
NES
NONSESE
codon
de
pa
func
ón
o
e
ES
M
SSENSE
ro
A
A
d
f
EVAT
VA
camen
e
s
m
SERVAT
VA
a
emp
azaarada
expres
ón
gén
feren
e
a
3
a
pos
ado
a
y
2dan
ca
c
ón
codon
pos
c
ón
coddon

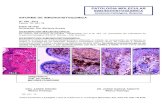
Gene No. of codons Nonsynonymous Synonymous
Histone H3 135 0.00 6.38
Histone H4 101 0 00 6 12Histone H4 101 0.00 6.12
Actin α 376 0.01 3.68
Aldolase A 363 0.07 3.59
HPRT 217 0.13 2.13
Insulin 51 0.13 4.02
α-Globin 141 0.55 5.14
β-Globin 144 0.80 3.05
Albumin 590 0.91 6.63
Ig VH 100 1.07 5.66
Growth hormone 189 1.23 4.95
Ig κ 106 1.87 5.66
Interferon-β1 159 2.21 5.88
Interferon-γ 136 2.79 8.59

•MUTACIONES QUE GENERAN GANANCIA O PERDIDA DE MATERIAL GENETICO•DNA CODIFICANTE Y NO CODIFICANTE•INVOLUCRA GENERALMENTE
•“REPETICIONES CORTAS EN TANDEM”• FENOMENOS DE RECOMBINACION
Short Tandem Repeats (STRs)AATGAATG
7 repeats
8 repeats


Homologous equal crossover Homologous equal crossover can result in fusion genes.The example shows how intragenic equal crossover occurring between alleles on occurring between alleles on nonsister chromatids can generate novel fusion genes composed of adjacent
t f th t segments from the two alleles. Note that similar exchanges between genes on sister chromatids do not
l i i l result in genetic novelty because the gene sequences on the interacting sister chromatids would be expected to be identical.

Unequal crossover and unequal Unequal crossover and unequal sister chromatid exchange cause insertions and deletions. The examples illustrate unequal pairing of chromatids within a tandemly
d U l repeated array. Unequal crossover involves unequal pairing of nonsister chromatids followed by chromatid breakage and rejoining. Unequal sister chromatid exchange Unequal sister chromatid exchange involves unequal pairing of sister chromatids followed by chromatidbreakage and rejoining. For the sake of simplicity, the breakages of the chromatids are shown to occur the chromatids are shown to occur between repeats, but of course breaks can occur within repeats. Note that both types of exchange are reciprocal - one of the
ti i ti h tid l participating chromatids loses some DNA, while the other gains some.

Unequal crossover in a tandem repeat Unequal crossover in a tandem repeat array can result in sequence homogenization. Note that the initial spread of the novel sequence variant to the same position in the chromosomes of other members of a sexual population p pcan result by random genetic drift. Once the mutation has achieved a reasonable population frequency (left panel) it can spread to other positions within the array (right panel). This can occur by successive gain of mutant repeats as a successive gain of mutant repeats as a result of unequal crossover (or unequal sister chromatid exchanges) and occasional loss of normal repeats. Eventually the mutant repeat can replace the original repeat sequence at replace the original repeat sequence at all positions within the array, leading to sequence homogenization for the mutant repeat. Such sequence homogenization is thought to result in species-specific concerted evolution for repetitive DNA
UEC l sequences. UEC, unequal crossover.

Tandem gene duplication Tandem gene duplication can result from unequal crossover or unequal sister chromatid exchange, facilitated by short facilitated by short interspersed repeats. The double arrow indicates the extent of the tandem gene d li ti f t duplication of a segment containing gene A and flanking sequences. Original mispairing of chromatids
ld b f ili d b hi h could be facilitated by a high degree of sequence homology between nonallelic short repeats (R1, R2). Note that p ( 1, 2)the same mechanism can result in large-scale deletions.

Gene conversion involves a nonreciprocalGene conversion involves a nonreciprocalsequence exchange between allelic ornonallelic genes. (A) Interallelic geneconversion. Note the nonreciprocal natureof the sequence exchange - the donorsequence is not altered but the acceptorq psequence is altered by incorporatingsequence copied from the donor sequence.(B) Interlocus gene conversion. This isfacilitated by a high degree of sequencehomology between nonallelic sequences, asin the case of tandem repeats (C) Mismatchin the case of tandem repeats. (C) Mismatchrepair of a heteroduplex. This is one ofseveral possible models to explain geneconversion. The model envisages invasion byone strand of the donor sequence (-) to forma heteroduplex with the complementary (+)a heteroduplex with the complementary ( )strand of the acceptor sequence, therebydisplacing the other strand of the acceptor.Mismatch repair enzymes recognize themispaired bases in the heteroduplex and‘correct' the mismatches so that the (+)
i ‘ d' bacceptor sequence is ‘converted' to beperfectly complementary in sequence to the(-) donor strand. Subsequent replication ofthe (-) acceptor strand and sealing of nicksresults in completion of the conversion.

LUGARES DE DNA
CARACTERISTICA
SECUENCIAS CODIFICANTES DEL GEN
•La mayoría patológicas (sustituciones) (1ra y 2da posicíón)GEN (sustituciones) (1ra y 2da posicíón)•Sitios mas susceptibles : islas CpGcerca a hotspotscerca a hotspots
SECUENCIAS INTRAGENIAS NO •Menor porcentaje mutacional
(10 15%)CODIFICANTES (10-15%)•Afecta regiones intronicas
SECUENCIAS REGULADORAS FUERA DE EXONES
•En las regiones promotoras de los genes (antes del Exon 1)g ( )•Alteración en la metilación


Mutations at conserved splice donor (SD) Mutations at conserved splice donor (SD) or splice acceptor (SA) sequences (see Figure 1.15 for consensus sequences) result in (A) intron retention where there is failure of splicing and an intervening intron sequence is not g qexcised; or in exon skipping where the spliceosome brings together the splice donor and splice acceptor sites of non-neighbouring exons. (B) Sequences that are very similar to the consensus splice donor or splice acceptor sequences may donor or splice acceptor sequences may coincidentally exist in introns and exons(sd and sa). These sequences are not normally used in splicing and so are known as cryptic splice sites. A mutation can activate a cryptic splice site by can activate a cryptic splice site by making the sequence more like the consensus splice donor or acceptor sequence and the cryptic splice site can now be recognized and used by the spliceosome (activation of the cryptic
li i ) S Fi 9 12 d 9 13splice site). See Figures 9.12 and 9.13for examples of activation of an exonicand an intronic cryptic splice site, respectively

GANANCIA DE INTRON
PERDIDA DE EXONDE EXON

GENERACION DE INSERCIONES O DELECIONES EN EL DNA PRODUCIENDO MUTACIONES TIPO “FRAMESHIFT”

Location and nature of mutation Effect on gene function CommentsExtragenic mutation Normally none Rare mutations may result in inactivation of distant regulatory elements required for normal
gene expression (see Figure 8.23)
Multigene deletion Abolition Associated with contiguous gene syndromes (see Figure 16.9)
Whole gene deletion AbolitionWhole gene duplication Can have effect due to altered gene
dosageLarge duplications including the peripheral myelin protein 22 gene can cause Charcot-Marie-Tooth syndrome (see Figure 16.7)
Whole exon deletion Abolition or modification May cause shift in reading frame; protein often unstable
Within exon Abolition If loss/change of key amino acids, shift of the reading frame or introduction of premature stopcodon
Modification If nonconservative substitutions, small in-frame insertions or other mutations at somelocations
None If conservative/silent substitutions or mutation at nonessential sites
Whole intron deletion NoneSplice site mutation Abolition or modulation of expression Conserved GT and AG signals are critically important for normal gene expression. Mutations
i d ki i i t t timay induce exon skipping or intron retention
Promoter mutation Abolition or modulation of expression Deletion, insertion or substitution of nucleotides within promoter may alter expression.Complete deletion abolishes function
Mutation of termination codon Modification Additional amino acids are included at the end of the protein until another stop codon isreached
Mutation of poly(A) signal Abolition or modulation of expression Deletion, insertion or substitution of nucleotides within poly(A) site may alter expression.Complete deletion abolishes functionComplete deletion abolishes function
Elsewhere in introns/UTS Usually none

Change Example
Delete:
(i) the entire gene Most α thalassemia mutations(i) the entire gene Most α‐thalassemia mutations
(ii) part of the gene 60% of Duchenne muscular dystrophy
Insert a sequence into the gene Insertion of LINE‐1 repetitive sequence into F8C gene in hemophilia A
Disrupt the gene structure:
(i) by a translocation X‐autosome translocations in women with Duchennemuscular dystrophy
(ii) by an inversion Inversion in F8C gene(ii) by an inversion Inversion in F8C gene
Prevent the promoter working:
(i) by mutation β‐Globin ‐29A → G mutation
(ii) by methylation Fragile‐X full mutation (FMR1)
Destabilize the mRNA:
(i) by a polyadenylation site mutation α‐globin AATAAA → AATAGA mutation
(ii) by nonsense‐mediated RNA decay Fibrillin mutations (FBN1)
Prevent correct splicing :Prevent correct splicing :
(i) by inactivating donor splice site PAX3 451 + 1G → T mutation
(ii) by inactivating acceptor splice site PAX3 452‐2A → G mutation
(iii) by activating a cryptic splice site β‐Globin intron 1 ‐110G → A mutation
Introduce a frameshift in translation PAX3 874–875insG mutation
Convert a codon into a stop codon PAX3 Q254X mutation
Replace an essential aminoacid PAX3 R271C mutation
Prevent post‐transcriptional processing Cleavage‐resistant collagen N‐terminal propeptide inPrevent post transcriptional processing Cleavage resistant collagen N terminal propeptide in Ehlers Danlos VII syndrome
Prevent correct cellular localization of product F508del mutation in cystic fibrosis


NOMENCLATURA EFECTO EN EL ALELO
ALELO NULO El alelo no genera prod ctoALELO NULO El alelo no genera producto proteico
A. HIPOMORFO Genera una reducida cantidad del producto
A.HIPERMORFO Genera una excesiva cantidad del producto p
A. NEOMORFO Genera un nuevo producto
A ANTIMORFO G d tA. ANTIMORFO Genera un producto que antagoniza la actividad o f ió d d tfunción de un producto normal

óSustitución a.a.R117H: arginina por hi tidi 117 (A 117
Deleciones –i ihistidina en 117 (Arg117
His) G542X: glicina por Stop
insercionesDelta - F508 o ∆F508
G542X: glicina por Stop (Gly Stop) Sustitución nucleótidos
nt6232(del5) o nt6232(del ACCTG) Sustitución nucleótidos
1162(G>A) 621(G>T)
( )nt409(insC)
( )

IVS4+1G>T :Cambio de G por T en la primera base del intron44
409-410insC :I ió d C l l id 409 410Inserción de C entre los nucleotidos 409 y 410

PERDIDA DE FUNCION
Enfermedades A.R.
HaploinsuficienciaA.D.
Efecto dominante negativo A.D. Toxicidad celularA.R.
< 50% de la actividad
A.D. negativo A.D.
Expansión de tripletas actividad
enzimaticatripletas
(GRANDES)
GANANCIA DE FUNCION
Proteínas hiperfuncionantes o
autoestimuladas
Proteínas neoformadas
Enfermedades A.D.
Repetición de tripletas con presencia de
proteínas con poli glutamina (MEDIANA)
ABL -BCL

Loss of function mutations in the PAX3 gene. The 10 exons of the gene are shown as boxes, with the connecting intronsnot to scale. The shaded areas show the sequences encoding the two DNA-binding domains of the PAX3 protein. Note that mutations that completely destroy the structure of the PAX3 protein (drawn above the gene diagram) are scattered that mutations that completely destroy the structure of the PAX3 protein (drawn above the gene diagram) are scattered over at least the first six exons of the gene, but missense mutations (shown below the gene diagram) are concentrated in two regions, the 5′ part of the paired domain and the third helix of the homeodomain. A196T is believed to affect splicing. The 874–875insG mutation introduces a seventh G into a run of six Gs; it has arisen independently several times and illustrates the relatively high frequency of slipped-strand mispairing

HPRT activity (% of normal) Phenotype>60 Normal8–60 Neurologically normal; hyperuricemia (gout)1.6–8 Neurological problem (choreoathetosis)1 4 1 6 h h d ( h h i lf1.4–1.6 Lesch-Nyhan syndrome (choreoathetosis, self-
mutilation) but intelligence normal<1.4 Classical Lesch-Nyhan syndrome (MIM 308000;y y ( ;
choreoathetosis, self-mutilation and mentalretardation)


DELECIONES E INSERCIONES CON
α GLOBINA (α talasemia) DELECION
FORMACON DE FRAMESHIFT
talase a) C ONFACTOR 8 (Hemofilia A) INSERCION
SUSTITUCIONES TIPO NONSENSE
INSERCIONDISTOFINA (Distrofia de Duchenne) MUTACIONES CON
ALTERACION EN EL
Duchenne) FRAMESHIFTPAX 3 (S W d b ) SPLICING PAX 3 (S. Waardenburg) I,D,S,Splic

Condition MIM no. Gene
Alagille syndrome
118450 JAG1
Multiple exostoses
133700 EXT1
Tomaculous 162500 PMP22Tomaculous neuropathy
162500 PMP22
Supravalvular aortic stenosis
185500 ELN
Tricho-rhino-phalangeal syndrome
190350 TRPS1
Waardenburg syndrome Type 1
193500 PAX3




Malfunction Gene Disease MIM no.
Overexpression PMP22 Charcot-Marie-Tooth 118200pdisease
Receptor permanently ‘on'
GNAS1 McCune-Albright disease
174800on disease
Acquire new substrate PI (Pittsburgh allele) α1-Antitrypsin deficiency
107400
Ion channel inappropriately open
SCN4A Paramyotonia congenita
168300
Structurally abnormal multimers
COL2A1 Osteogenesis imperfecta
Various
Protein aggregation HD Huntington disease 143100
Chimeric gene BCR-ABL Chronic myeloid 151410leukemia

Tumor Rearrangement Chimeric gene Nature of chimeric product
CML t(9;22)(q34;q11) BCR-ABL Tyrosine kinase
Ewing sarcoma t(11;22)(q24;q12) EWS-FLI1 Transcription factor
Ewing sarcoma (variant)
t(21;22)(q22;q12) EWS-ERG Transcription factor( )Malignant melanoma of soft parts
t(12;22)(q13;q12) EWS-ATF1 Transcription factor
Desmoplastic small round cell tumor
t(11;22)(p13;q12) EWS-WT1 Transcription factor
Liposarcoma t(12;16)(q13;p11) FUS-CHOP Transcription factor
AML t(16;21)(p11;q22) FUS-ERG Transcription factor
Papillary thyroid carcinoma
inv(1)(q21;q31) NTRK1-TPM3(TRKoncogene)
Tyrosine kinasecarcinoma oncogene)
Pre-B cell ALL t(1;19)(q23;p13.3) E2A-PBX1 Transcription factor
ALL t(X;11)(q13;q23) MLL-AFX1 Transcription factor
ALL T(4;11)(q21;q23) MLL-AF4 Transcription factor
ALL t(9;11)(q21;q23) MLL-AF9 Transcription factor
ALL t(11;19)(q23;p13) MLL-ENL Transcription factor
Acute promyelocytic leukemia
t(15;17)(q22;q12) PML-RARA Transcriptionfactor+retinoic acidleukemia factor+retinoic acidreceptor
Alveolar rhabdomyosarcoma
t(2;13)(q35;q14) PAX3-FKHR Transcription factor

Rearreglos cromosómicos con producción de genes quiméricos

Gene Location Diseases Symbol MIM no.PAX3 2q35 Waardenburg syndrome type 1 WS1 193500
Alveolar rhabdomyosarcoma RMS2 268220
CFTR 7p31.2 Cystic fibrosis CF 219700Bilateral absence of vas deferens
RET 10q11.2 Multiple endocrine neoplasia type 2A MEN2A 171400
Multiple endocrine neoplasia type 2B MEN2B 162300
Medullary thyroid carcinoma FMTC 155420Hirschsprung disease HSCR 142623p g
PMP22 17p11.2 Charcot-Marie-Tooth neuropathy type 1A CMT1A 118220
Tomaculous neuropathy HNPP 162500SCN4A 17q23.1-q25.3 Paramyotonia congenita PMC 168300
Hyperkalemic periodic paralysis HYPP 170500Acetazolamide-responsive myotonia congenita
PRNP 20p12 pter Creutzfeldt Jakob disease CJD 123400PRNP 20p12-pter Creutzfeldt-Jakob disease CJD 123400Familial fatal insomnia FFI 176640
GNAS1 20q13.2 Albright hereditary osteodystrophy AHO 103580
McCune Albright syndrome PFD 174800McCune-Albright syndrome PFD 174800AR Xcen-q22 Testicular feminization syndrome TFM 313700
Kennedy disease SBMA 313200

Gran heterogeneidad genéticaCMT
PMP22, peripheral myelinprotein 22MPZ m elin protein eroMPZ, myelin protein zeroGJB1, connexin 32EGR2, early growth response proteinproteinNDRG1, N-myc down regulated gene 1,MTMR2, myotubularin relatedMTMR2, myotubularin relatedprotein 2 kinaseGAN1, gigaxoninNEFL, neurofilament light chainKIF1B, kinesin 1BβPRX, periaxin

Gene Location Mutations Syndrome
COL1A1 17 22 N ll ll l OI t ICOL1A1 17q22 Null alleles OI type I
Partial deletions; C-terminalsubstitutions
OI type II
N terminal s bstit tions OI t pes I III or IVN-terminal substitutions OI types I, III or IV
Deletion of exon 6 EDS type VII
COL1A2 7q22.1 Splice mutations; exon deletions OI type I
C-terminal mutations OI type II, IV
N-terminal substitutions OI type III
D l ti f 6 EDS t VIIDeletion of exon 6 EDS type VII
COL2A1 12q13 Point mutations SED
Nonsense mutation Stickler syndrome
f i i i d l iDefect in conversion Kniest dysplasia
Missense Achondrogenesis II, spondylo-meta-epiphyseal dysplasia
COL11A2 6p21.3 Splicing mutation Stickler syndrome